South African company launches AI-based Covid-19 risk screening mobile app
South African company UC-Wireless has launched a new artificial intelligence- (AI-) driven mobile application to provide advanced healthcare risk wellness and Covid-19 screening for business staff, clients and personal users.
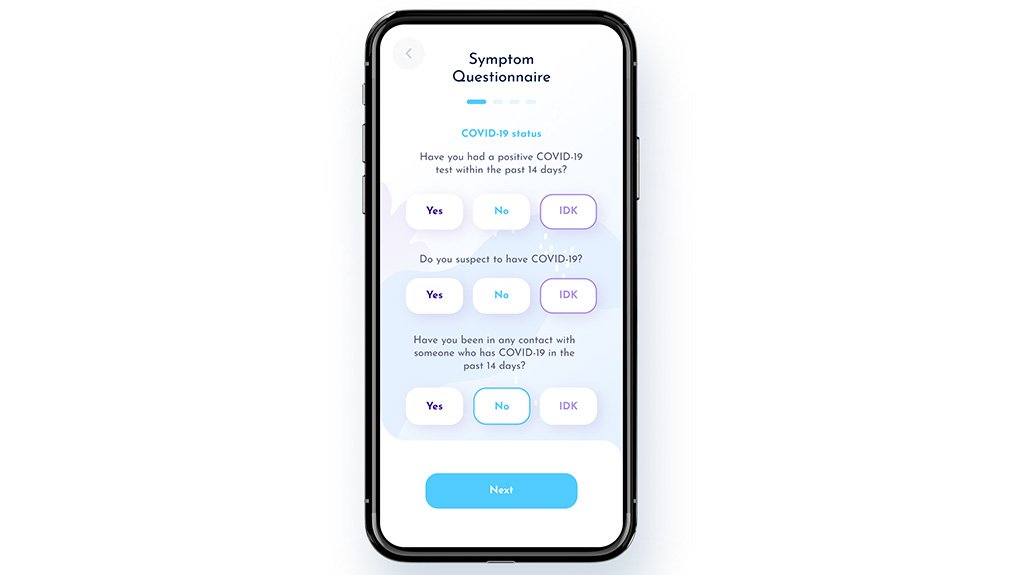
The smart Instant Vitals app uses the mobile phone’s camera and AI integrated within the application to screen for the main recognised symptoms of Covid-19 and measure vital signs, including oxygen saturation, heart rate and respiratory rate.
This, in effect, turns mobile devices into an easy-to-use clinical tool exhibiting an acceptable standard of medical device accuracy.
Several studies, reviews and reports are starting to show that temperature measurements alone has its limitations in the detection of Covid-19, as temperature and fever often do not exhibit, present late in the progression of the infection or patients are asymptomatic and do not show any symptoms.
“There is proof that the critical early warning signs of Covid-19 include a reduction in oxygen saturation (below 95%), and/or a high resting heart rate (above 100 beats per minute), or an increase in respiratory rate (above 20 breaths per minute) in a resting person,” the company explained.
While measuring temperature remains important, there is a need for the advanced screening provided by Instant Vitals, which was designed and developed in collaboration with medical doctors, engineers, professors and digital and technology experts.
The app boasts an integrated QR code mechanism to manage access to businesses and venues, and extracts vital signs with good clinical lab-tested accuracy within 20 to 50 seconds by processing video which the AI detects from skin regions of the face.
Instant Vitals generates a unique encrypted QR code for each person’s device for screening once individual and organisation licences are purchased and registered on the app.
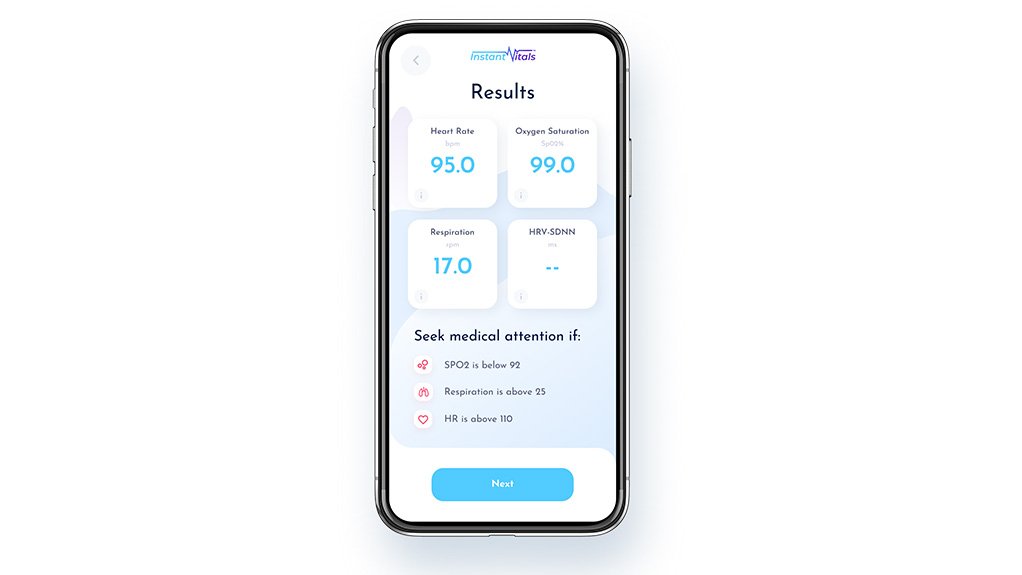
When the QR code is scanned and the symptom questionnaire completed, the vital signs are measured.
“It runs like any other app on a mobile phone or tablet. The app calculates a risk status from the vital signs and the answers to the screening questions, which are encrypted and stored in the Instant Vitals secure cloud service for each person,” the company said in a statement.
The app is Protection of Personal Information Act- and European Union General Data Protection Regulation-compliant, with the information captured kept private.
The only data made available publicly is the person’s risk status at the point of entry when their QR code or mobile number for visitors is provided for screening.
The organisations using Instant Vitals for their business premises can see risk status reporting on their app’s dashboard.
The risk status of screened individuals is flagged as green, indicating healthy; amber, indicating caution as vital signs or symptoms are not normal but not high risk; or red, where one or more vital signs or symptoms are indicating a risk which should be referred for medical attention or diagnosis.
The app also shows a critical status, which is an extreme red status and means urgent medical treatment should be sought by the individual, based on the vital signs measured and symptoms analysed by the app.
Those whose risk results are red or critical status should be referred for Covid-19 testing.
“The app caters for mass QR code scanning by individuals who screened at home. It also enables screening for visitors and staff without phones at corporate offices, restaurants, retail stores, hospitals, clinics, pharmacies, public transport facilities, schools, universities, airports, industrial sites and mines, among others.”
Two licence options – a personal app and business screening verifier app, including the dashboard – are available from the Apple iStore and Google Play store.
The licences will initially be available through resellers and partners of UC-Wireless across Africa and globally.
While UC-Wireless has been registered with the South African Health Products Regulatory Authority as a manufacturer incorporating the certified AI and the app Instant Vitals, the app is not intended to replace medical advice, therefore individuals should consult with qualified health care professionals for diagnosis and advice.
The licensed technology meets the requirements of global accuracy regulations and applications for approvals from the US-based Food and Drug Administration and the European CE mark are in progress.
Article Enquiry
Email Article
Save Article
Feedback
To advertise email advertising@creamermedia.co.za or click here
Press Office
Announcements
What's On
Subscribe to improve your user experience...
Option 1 (equivalent of R125 a month):
Receive a weekly copy of Creamer Media's Engineering News & Mining Weekly magazine
(print copy for those in South Africa and e-magazine for those outside of South Africa)
Receive daily email newsletters
Access to full search results
Access archive of magazine back copies
Access to Projects in Progress
Access to ONE Research Report of your choice in PDF format
Option 2 (equivalent of R375 a month):
All benefits from Option 1
PLUS
Access to Creamer Media's Research Channel Africa for ALL Research Reports, in PDF format, on various industrial and mining sectors
including Electricity; Water; Energy Transition; Hydrogen; Roads, Rail and Ports; Coal; Gold; Platinum; Battery Metals; etc.
Already a subscriber?
Forgotten your password?
Receive weekly copy of Creamer Media's Engineering News & Mining Weekly magazine (print copy for those in South Africa and e-magazine for those outside of South Africa)
➕
Recieve daily email newsletters
➕
Access to full search results
➕
Access archive of magazine back copies
➕
Access to Projects in Progress
➕
Access to ONE Research Report of your choice in PDF format
RESEARCH CHANNEL AFRICA
R4500 (equivalent of R375 a month)
SUBSCRIBEAll benefits from Option 1
➕
Access to Creamer Media's Research Channel Africa for ALL Research Reports on various industrial and mining sectors, in PDF format, including on:
Electricity
➕
Water
➕
Energy Transition
➕
Hydrogen
➕
Roads, Rail and Ports
➕
Coal
➕
Gold
➕
Platinum
➕
Battery Metals
➕
etc.
Receive all benefits from Option 1 or Option 2 delivered to numerous people at your company
➕
Multiple User names and Passwords for simultaneous log-ins
➕
Intranet integration access to all in your organisation