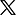SAHPRA registers Janssen Covid-19 vaccine with conditions
The South African Health Products Regulatory Authority (SAHPRA) announced on Monday the registration of the Janssen Covid-19 vaccine on March 31, with conditions.
SAHPRA CEO Dr Boitumelo Semete-Makokotlela explained that one of the conditions is that the vaccine be manufactured under conditions of Good Manufacturing Practices (GMP) as determined by SAHPRA and aligned with global best practice.
SAHPRA will seek documentation from the regulatory authority in the country manufacturing the vaccine.
Meanwhile, the SAHPRA is in discussion with the US Food and Drug Administration (FDA) around concerns with a non-compliant batch of vaccines.
This has led to an investigation of more batches by the US FDA.
“…and SAHPRA is awaiting reports from the US FDA on whether or not these other batches were manufactured according to GMP standards and if the batches are contaminated. Until the FDA has shared these reports, SAHPRA has insufficient information to approve specific batches of the Janssen Covid-19 vaccine,” the SAHPRA said in a statement.
Article Enquiry
Email Article
Save Article
Feedback
To advertise email advertising@creamermedia.co.za or click here
Press Office
Announcements
What's On
Subscribe to improve your user experience...
Option 1 (equivalent of R125 a month):
Receive a weekly copy of Creamer Media's Engineering News & Mining Weekly magazine
(print copy for those in South Africa and e-magazine for those outside of South Africa)
Receive daily email newsletters
Access to full search results
Access archive of magazine back copies
Access to Projects in Progress
Access to ONE Research Report of your choice in PDF format
Option 2 (equivalent of R375 a month):
All benefits from Option 1
PLUS
Access to Creamer Media's Research Channel Africa for ALL Research Reports, in PDF format, on various industrial and mining sectors
including Electricity; Water; Energy Transition; Hydrogen; Roads, Rail and Ports; Coal; Gold; Platinum; Battery Metals; etc.
Already a subscriber?
Forgotten your password?
Receive weekly copy of Creamer Media's Engineering News & Mining Weekly magazine (print copy for those in South Africa and e-magazine for those outside of South Africa)
➕
Recieve daily email newsletters
➕
Access to full search results
➕
Access archive of magazine back copies
➕
Access to Projects in Progress
➕
Access to ONE Research Report of your choice in PDF format
RESEARCH CHANNEL AFRICA
R4500 (equivalent of R375 a month)
SUBSCRIBEAll benefits from Option 1
➕
Access to Creamer Media's Research Channel Africa for ALL Research Reports on various industrial and mining sectors, in PDF format, including on:
Electricity
➕
Water
➕
Energy Transition
➕
Hydrogen
➕
Roads, Rail and Ports
➕
Coal
➕
Gold
➕
Platinum
➕
Battery Metals
➕
etc.
Receive all benefits from Option 1 or Option 2 delivered to numerous people at your company
➕
Multiple User names and Passwords for simultaneous log-ins
➕
Intranet integration access to all in your organisation