Fast-tracking construction of biotech facilities

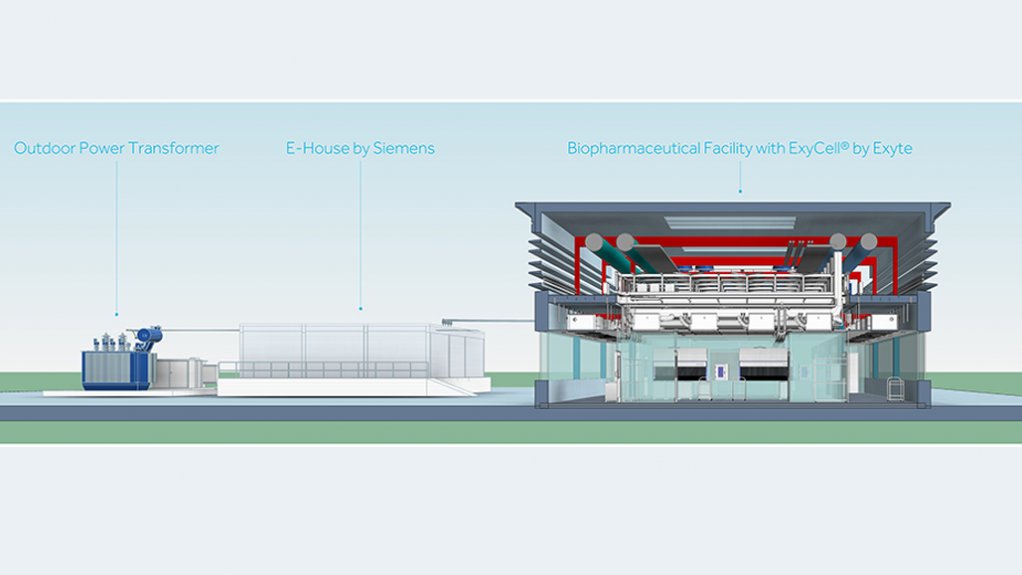
Model of a biopharmaceutical facility consisting of pre-configured ExyCell modules and an integrated power supply by Siemens
The Covid-19 pandemic poses major challenges for pharmaceutical companies, specifically related to the development and mass-production of new vaccines and medicines in existing production facilities.
A new partnership between Siemens and Exyte aims to offer end-to-end solutions for the biopharmaceutical industry. The two companies offer standardised, turnkey solutions compliant with current Good Manufacturing Practice and Good Automated Manufacturing Practice that are specifically designed for the needs of biotechnology manufacturers, as well as cell and gene-therapy manufacturers.
“We are excited to collaborate with Exyte to provide pre-fabricated, modular biotechnology solutions with our technology already embedded. As a market leader for the pharmaceutical industry, we offer know-how for process automation with our Simatic PCS 7 and WinCC automation platforms, as well as power supply and fire protection for modular cleanrooms,” enthuses Siemens.
Currently, the first buildings with Siemens’ technology and ExyCell modules are being designed for cell and gene therapy manufacturing and biologicals production in China and Europe.
Exyte states: “Our partnership with Siemens enables us to pre-integrate Siemens’ technology into ExyCell modules, thus offering clients end-to-end solutions for their facilities. Our collaboration allows our clients to reap the benefits of Industry 4.0 without the necessity of engineering them from scratch each time, thus saving them time and money. ExyCell modules are suitable for new buildings as well as for the retrofit of existing buildings, and can be provided either in standard, off-the-shelf plant configurations, or modules that can be combined to meet specific customer requirements.”
Article Enquiry
Email Article
Save Article
Feedback
To advertise email advertising@creamermedia.co.za or click here
Announcements
What's On
Subscribe to improve your user experience...
Option 1 (equivalent of R125 a month):
Receive a weekly copy of Creamer Media's Engineering News & Mining Weekly magazine
(print copy for those in South Africa and e-magazine for those outside of South Africa)
Receive daily email newsletters
Access to full search results
Access archive of magazine back copies
Access to Projects in Progress
Access to ONE Research Report of your choice in PDF format
Option 2 (equivalent of R375 a month):
All benefits from Option 1
PLUS
Access to Creamer Media's Research Channel Africa for ALL Research Reports, in PDF format, on various industrial and mining sectors
including Electricity; Water; Energy Transition; Hydrogen; Roads, Rail and Ports; Coal; Gold; Platinum; Battery Metals; etc.
Already a subscriber?
Forgotten your password?
Receive weekly copy of Creamer Media's Engineering News & Mining Weekly magazine (print copy for those in South Africa and e-magazine for those outside of South Africa)
➕
Recieve daily email newsletters
➕
Access to full search results
➕
Access archive of magazine back copies
➕
Access to Projects in Progress
➕
Access to ONE Research Report of your choice in PDF format
RESEARCH CHANNEL AFRICA
R4500 (equivalent of R375 a month)
SUBSCRIBEAll benefits from Option 1
➕
Access to Creamer Media's Research Channel Africa for ALL Research Reports on various industrial and mining sectors, in PDF format, including on:
Electricity
➕
Water
➕
Energy Transition
➕
Hydrogen
➕
Roads, Rail and Ports
➕
Coal
➕
Gold
➕
Platinum
➕
Battery Metals
➕
etc.
Receive all benefits from Option 1 or Option 2 delivered to numerous people at your company
➕
Multiple User names and Passwords for simultaneous log-ins
➕
Intranet integration access to all in your organisation